In a significant advancement at the intersection of artificial intelligence and molecular biology, researchers have developed BioBridge—a novel framework that bridges the gap between specialized protein language models and general-purpose large language models. Published on arXiv as a preprint (arXiv:2602.17680), this innovative approach addresses fundamental limitations in both types of models to create a more versatile and capable system for biological reasoning.
The Protein-Language Divide
Protein Language Models (PLMs) have revolutionized computational biology by learning representations of protein sequences that capture structural and functional properties. These specialized models excel at predicting protein functions, interactions, and properties but suffer from limited adaptability across diverse biological contexts and poor generalization to tasks beyond their training scope.
Conversely, general-purpose Large Language Models (LLMs) demonstrate remarkable reasoning capabilities and broad knowledge but lack the specialized understanding required to interpret protein sequences and perform domain-specific biological reasoning. This fundamental divide has created a bottleneck in AI-driven biological discovery.
The BioBridge Architecture
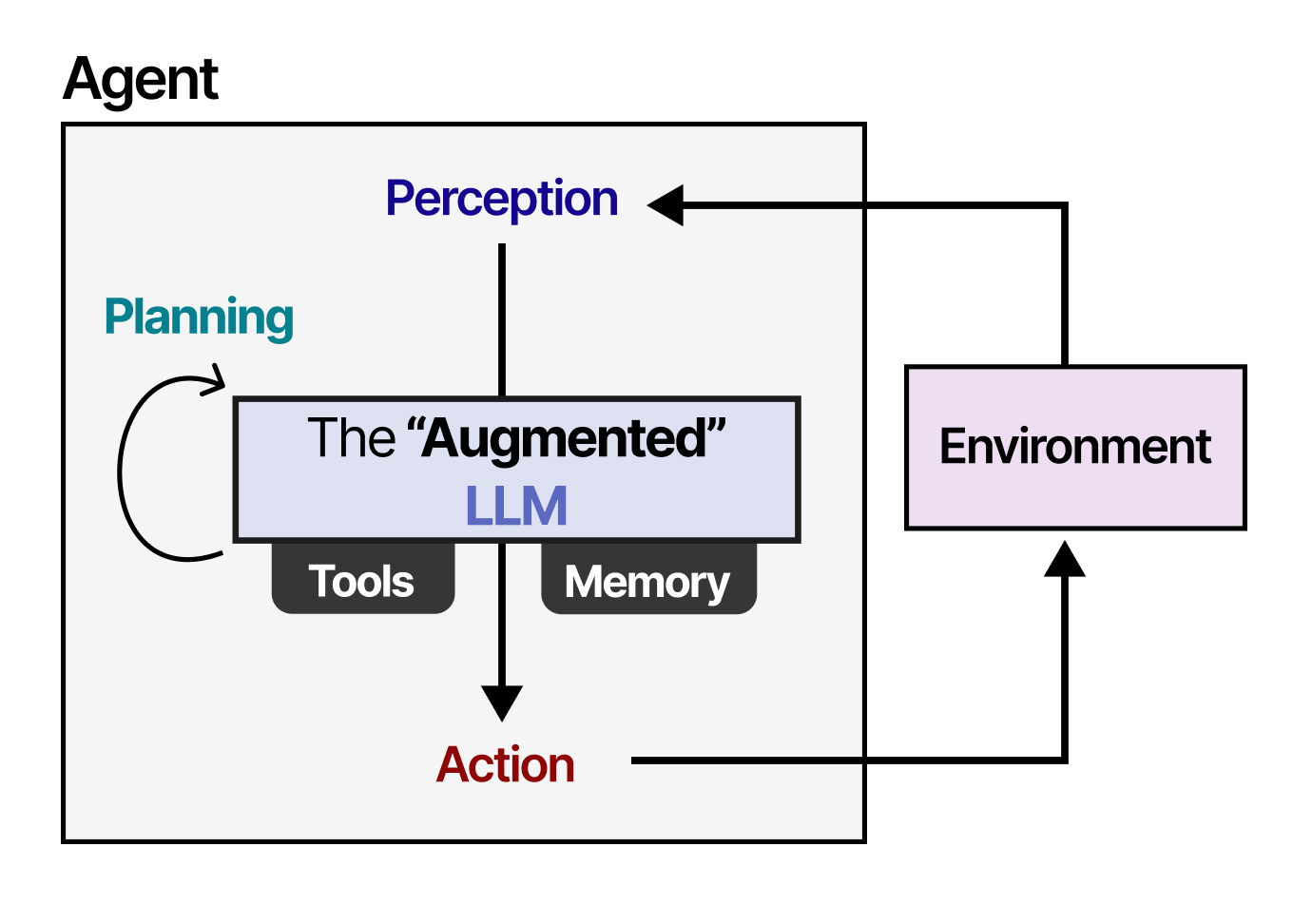
BioBridge employs a sophisticated three-component architecture designed to overcome these limitations:
1. Domain-Incremental Continual Pre-training (DICP): This innovative approach simultaneously infuses protein domain knowledge and general reasoning corpus into an LLM, effectively mitigating catastrophic forgetting—the tendency of neural networks to forget previously learned information when trained on new tasks.
2. Cross-Modal Alignment Pipeline: The system implements a PLM-Projector-LLM pipeline that maps protein sequence embeddings from specialized protein models into the semantic space of the language model. This creates a shared representation space where protein information can be processed alongside natural language.
3. End-to-End Optimization: BioBridge supports various tasks through unified optimization, including protein property prediction and knowledge question-answering, creating a versatile system applicable across multiple biological research domains.
Performance Breakthroughs
The research team reports that BioBridge demonstrates performance comparable to mainstream PLMs on multiple protein benchmarks, including Enzyme Commission (EC) classification and BindingDB for protein-ligand interactions. Simultaneously, it achieves results on par with LLMs on general understanding tasks like MMLU (Massive Multitask Language Understanding) and RACE (Reading Comprehension Dataset).
This dual capability represents a significant advancement over existing approaches. Traditional PLMs typically sacrifice general language understanding for specialized protein knowledge, while LLMs lack the domain specificity required for sophisticated biological reasoning. BioBridge appears to have broken this trade-off.
Implications for Biological Research
The development of BioBridge has profound implications for multiple areas of biological research:
Drug Discovery: By enabling more sophisticated reasoning about protein-ligand interactions and protein functions, BioBridge could accelerate the identification of potential drug targets and therapeutic compounds.
Synthetic Biology: The system's enhanced understanding of protein sequences and functions could support the design of novel proteins with specific properties for industrial, medical, or environmental applications.
Personalized Medicine: Improved protein understanding combined with reasoning capabilities could enhance the interpretation of genetic variations and their implications for individual health outcomes.
Scientific Communication: BioBridge's dual proficiency in specialized biological knowledge and general language could facilitate more effective communication between domain experts and broader scientific communities.
Technical Innovations and Challenges
The BioBridge framework introduces several technical innovations worth noting. The Domain-Incremental Continual Pre-training approach represents an advancement in continual learning techniques, particularly for combining highly specialized and general knowledge domains. The cross-modal alignment mechanism demonstrates how information from fundamentally different data types (protein sequences and natural language) can be effectively integrated.
However, the researchers acknowledge several challenges that remain. The computational requirements for training such hybrid systems are substantial, potentially limiting accessibility for smaller research institutions. Additionally, while the system shows promising results on established benchmarks, real-world biological problems often present more complex, multifaceted challenges that may require further refinement.
Future Directions
The BioBridge team suggests several promising directions for future research. These include expanding the framework to incorporate additional biological data types (such as gene expression data or metabolic pathways), developing more efficient training methodologies to reduce computational costs, and exploring applications in emerging areas like protein design and evolutionary biology.
The preprint nature of the publication on arXiv—an open-access repository for scientific papers before formal peer review—means the research community will now engage in rigorous evaluation and validation of these findings. This process typically involves independent testing, methodological scrutiny, and attempts to reproduce reported results.
The Broader AI Landscape
BioBridge represents a growing trend in AI research toward creating hybrid systems that combine specialized domain knowledge with general reasoning capabilities. Similar approaches are emerging in other scientific domains, including materials science, climate modeling, and astrophysics, suggesting a paradigm shift in how AI systems are designed for scientific discovery.
The successful integration of protein language models with general-purpose LLMs also has implications for the future of multimodal AI systems. As researchers develop techniques to combine increasingly diverse data types and knowledge domains, we may see the emergence of more versatile and capable AI systems across scientific disciplines.
Source: arXiv:2602.17680, "BioBridge: Bridging Proteins and Language for Enhanced Biological Reasoning with LLMs" (Submitted February 4, 2026)