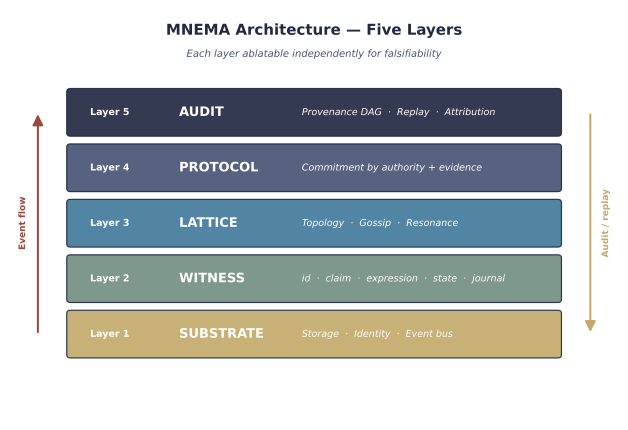
A wireless brain implant with 544 electrodes has been successfully implanted in a third blind human patient, bypassing eyes and optic nerves entirely. The device treats vision as a software problem, stimulating the visual cortex directly to create artificial sight.
Key facts
- Third successful human implantation of wireless visual cortex implant.
- 544 electrodes stimulate visual cortex directly, bypassing eyes and optic nerves.
- Device is fully wireless, no external wires or connectors.
- Prior comparable device (Orion) used 60 electrodes; this uses 544.
- Treats vision as software problem, not biological repair.
A wireless brain implant that completely bypasses the eyes and optic nerves has achieved its third successful human implantation, according to a post by @kimmonismus on X. The device uses 544 electrodes to stimulate the visual cortex directly, creating artificial sight for individuals with total blindness.
Instead of attempting to repair damaged eyes or optic nerves, the system treats vision as a software problem, patching visual information straight into the brain's hardware. This approach fundamentally redefines blindness treatment, shifting from biological repair to neural interface.
The implant is fully wireless, eliminating infection risks and mobility constraints associated with wired brain-computer interfaces. The company behind the device has not been named in the source, nor have specific patient outcomes or visual acuity metrics been disclosed.
Why This Matters
The unique take here is that this approach reframes blindness as a resolvable input/output problem rather than an incurable biological condition. If the 544-electrode array can produce usable visual perception—even low-resolution phosphene-based vision—it could obsolete decades of research into retinal implants and gene therapies, which require functional optic nerves.
Prior work in this space includes the Orion cortical visual prosthesis by Second Sight Medical Products, which was tested in six patients before the company ceased operations in 2020. That device used 60 electrodes. The new implant's 544 electrodes represent a 9x increase in resolution potential, though no direct comparison has been provided by the source.
Limitations
The source provides no details on patient demographics, implantation duration, adverse events, or the specific company or research group conducting the trials. The visual acuity achieved remains unspecified. Blindness treatment trials face high regulatory hurdles; a third successful implant suggests Phase I safety data may be accumulating, but efficacy endpoints remain unknown.
Wireless power transmission through the skull for a 544-electrode array raises engineering questions about heat dissipation and signal fidelity that have not been addressed in the source material.
What to watch
Watch for the release of peer-reviewed clinical data, visual acuity metrics (e.g., Snellen equivalent or grating acuity), and the identity of the company or research group. The next milestone is a pivotal trial with 10+ patients, likely within 12-18 months.